Numerical Simulation Study of Wound Remodeling
-
摘要:
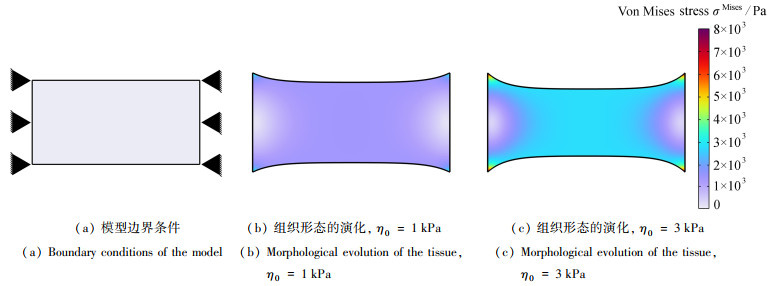
组织重塑普遍存在于人体的组织和器官,与形态发生、伤口愈合、纤维化发展以及癌症的扩散与转移等密切相关. 力学微环境在组织重塑过程中发挥重要作用,但是,目前张力调控影响创口重塑的规律仍不清. 该文建立了细胞主动收缩引起组织重塑的动态数理模型,并通过有限元方法模拟了不同预张力牵张组织中侧边和内部创口重塑的过程;进一步,基于该模型研究了张力调控对创口重塑的影响. 结果表明,张力调控显著影响创口重塑过程,通过适当的减张作用可以有效降低创口重塑过程中的应力水平和创口大小. 该研究有助于加深人们对于组织重塑过程中力学作用的理解,为从力学角度干预创口重塑过程提供了有用参考.
Abstract:The tissue remodeling is widespread in human tissues and organs, closely associated with morphogenesis, wound healing, fibrosis development, as well as cancer spread and metastasis. The mechanical microenvironment plays a crucial role in the tissue remodeling, yet the impact of tension regulation on wound remodeling remains unclear. A dynamic mathematical model for tissue remodeling induced by active cell contraction was established. The processes of lateral and inner wound remodeling in tissues with different pretensions were simulated with the finite element method. Additionally, the effects of tension regulation on wound remodeling were studied based on the model. The results show that, the tension regulation significantly influences the wound remodeling process, an appropriate tension reduction would effectively decrease the stress magnitude and the wound size. This study contributes to a deeper understanding of the mechanical effects in tissue remodeling and provides references for potential interventions in wound healing.
-
Key words:
- tissue remodeling /
- wound healing /
- finite element method /
- mechanical regulation
edited-byedited-by1) (我刊青年编委黄国友来稿) -
表 1 数值模拟参数
Table 1. simulation parameters
parameter symbol value Mooney-Rivlin parameter 1 C01/Pa 2 000 Mooney-Rivlin parameter 2 C10/Pa -500 initial bulk modulus κ/Pa 400 density ρ/(kg/m3) 1 000 maximum cell contractility η0/Pa 300~3 000 dimensionless function parameter ζ 0.5 boundary displacement u0/μm 20~200 initial length of wound lw/mm 0.14 -
[1] PINET K, MCLAUGHLIN K A. Mechanisms of physiological tissue remodeling in animals: manipulating tissue, organ, and organism morphology[J]. Developmental Biology, 2019, 451(2): 134-145. doi: 10.1016/j.ydbio.2019.04.001 [2] TABER L A. Biomechanics of growth, remodeling, and morphogenesis[J]. Applied Mechanics Reviews, 1995, 48: 487-545. doi: 10.1115/1.3005109 [3] KELLER R, DAVIDSON L A, SHOOK D R. How we are shaped: the biomechanics of gastrulation[J]. Differentiation, 2003, 71(3): 171-205. doi: 10.1046/j.1432-0436.2003.710301.x [4] BAN E, FRANKLIN J M, NAM S, et al. Mechanisms of plastic deformation in collagen networks induced by cellular forces[J]. Biophysical Journal, 2018, 114(2): 450-461. doi: 10.1016/j.bpj.2017.11.3739 [5] BALESTRINI J L, BILLIAR K L. Magnitude and duration of stretch modulate fibroblast remodeling[J]. Journal of Biomechanical Engineering, 2009, 131(5): 051005. doi: 10.1115/1.3049527 [6] BAINBRIDGE P. Wound healing and the role of fibroblasts[J]. Journal of Wound Care, 2013, 22(8): 407-408, 410-412. doi: 10.12968/jowc.2013.22.8.407 [7] SAKAR M S, EYCKMANS J, PIETERS R, et al. Cellular forces and matrix assembly coordinate fibrous tissue repair[J]. Nature Communications, 2016, 7: 11036. doi: 10.1038/ncomms11036 [8] CORR D T, HART D A. Biomechanics of scar tissue and uninjured skin[J]. Advances in Wound Care, 2012, 2(2): 37-43. [9] DESHPANDE V S, MCMEEKING R M, EVANS A G. A bio-chemo-mechanical model for cell contractility[J]. Proceedings of the National Academy of Sciences, 2006, 103(38): 14015-14020. doi: 10.1073/pnas.0605837103 [10] DALLON J C, EVANS E J, EHRLICH H P. A mathematical model of collagen lattice contraction[J]. Journal of The Royal Society Interface, 2014, 11(99): 20140598. doi: 10.1098/rsif.2014.0598 [11] LIU L, YU H, ZHAO H, et al. Matrix-transmitted paratensile signaling enables myofibroblast-fibroblast cross talk in fibrosis expansion[J]. Proceedings of the National Academy of Sciences of the United States of America, 2020, 117(20): 10832-10838. [12] HIRASHIMA T, RENS E G, MERKS R M H. Cellular Potts modeling of complex multicellular behaviors in tissue morphogenesis[J]. Development, Growth & Differentiation, 2017, 59(5): 329-339. [13] TSINGOS E, BAKKER B H, KEIJZER K A E, et al. Hybrid cellular Potts and bead-spring modeling of cells in fibrous extracellular matrix[J]. Biophysical Journal, 2023, 122(13): 2609-2622. doi: 10.1016/j.bpj.2023.05.013 [14] LOERAKKER S, OBBINK-HUIZER C, BAAIJENS F P T. A physically motivated constitutive model for cell-mediated compaction and collagen remodeling in soft tissues[J]. Biomechanics and Modeling in Mechanobiology, 2014, 13(5): 985-1001. doi: 10.1007/s10237-013-0549-1 [15] RODRIGUEZ E K, HOGER A, MCCULLOCH A D. Stress-dependent finite growth in soft elastic tissues[J]. Journal of Biomechanics, 1994, 27(4): 455-467. doi: 10.1016/0021-9290(94)90021-3 [16] KIM J, MAILAND E, ANG I, et al. A model for 3D deformation and reconstruction of contractile microtissues[J]. Soft Matter, 2021, 17(45): 10198-10209. doi: 10.1039/D0SM01182G [17] KIM J, MAILAND E, SAKAR M S, et al. A model for mechanosensitive cell migration in dynamically morphing soft tissues[J]. Extreme Mechanics Letters, 2023, 58: 101926. doi: 10.1016/j.eml.2022.101926 [18] BROWN R A, PRAJAPATI R, MCGROUTHER D A, et al. Tensional homeostasis in dermal fibroblasts: mechanical responses to mechanical loading in three-dimensional substrates[J]. Journal of Cellular Physiology, 1998, 175(3): 323-332. doi: 10.1002/(SICI)1097-4652(199806)175:3<323::AID-JCP10>3.0.CO;2-6 [19] WAHLSTEN A, STRACUZZI A, LVCHTEFELD I, et al. Multiscale mechanical analysis of the elastic modulus of skin[J]. Acta Biomaterialia, 2023, 170: 155-168. doi: 10.1016/j.actbio.2023.08.030 [20] LEGANT W R, PATHAK A, YANG M T, et al. Microfabricated tissue gauges to measure and manipulate forces from 3D microtissues[J]. Proceedings of the National Academy of Sciences, 2009, 106(25): 10097-10102. doi: 10.1073/pnas.0900174106 [21] THEOCHARIS A D, SKANDALIS S S, GIALELI C, et al. Extracellular matrix structure[J]. Advanced Drug Delivery Reviews, 2016, 97: 4-27. doi: 10.1016/j.addr.2015.11.001 [22] TOMASEK J J, GABBIANI G, HINZ B, et al. Myofibroblasts and mechano-regulation of connective tissue remodelling[J]. Nature Reviews Molecular Cell Biology, 2002, 3(5): 349-363. doi: 10.1038/nrm809 [23] MAILAND E, LI B, EYCKMANS J, et al. Surface and bulk stresses drive morphological changes in fibrous microtissues[J]. Biophysical Journal, 2019, 117(5): 975-986. doi: 10.1016/j.bpj.2019.07.041 [24] FRIEDMAN A, HU B, XUE C. A three dimensional model of wound healing: analysis and computation[J]. Discrete and Continuous Dynamical Systems: B, 2012, 17: 2691-2712. doi: 10.3934/dcdsb.2012.17.2691 [25] BAI J, ZENG X. Computational modeling and simulation of epithelial wound closure[J]. Scientific Reports, 2023, 13(1): 6265. doi: 10.1038/s41598-023-33111-4 [26] ZHANG S, NABI O, JIANG X. New strategy of modulating incision tension: a wound tension offloading device applied before surgery[J]. Dermatol Ther, 2021, 34(2): e14797. [27] 温广全, 纪小刚, 段玉顺, 等. 考虑材料参数不确定性的皮肤伤口缝合力预测模型[J]. 应用数学和力学, 2023, 44(4): 441-449. doi: 10.21656/1000-0887.430067?viewType=HTMLWEN Guangquan, JI Xiaogang, DUAN Yushun, et al. A prediction model for skin wound suture forces with uncertain material parameters[J]. Applied Mathematics and Mechanics, 2023, 44(4): 441-449. (in Chinese) doi: 10.21656/1000-0887.430067?viewType=HTML -




 下载:
下载:









 渝公网安备50010802005915号
渝公网安备50010802005915号